Troponin I and T are the main biomarkers and gold standards for diagnostics of acute myocardial infarction (AMI). Recently high- sensitivity cTn (hs-cTn) assays, the detection limit of which is pg/ml (ng/l) rather than ng/ml, have made it possible to identify any myocardial injury, including those AMI patients, within 1 to 3 hours. This represents a potential 3-hour time saving to ensure more rapid patient management.
The current generation of commercially available hs-cTn assays are approximately 1,000 times more analytically sensitive than the first cTnI assays. The hs-cTn assays can detect minor cardiac injury events from a long list of pathologies that cause myocardial tissue necrosis or cell death. For the development of hs-cTn assays we have developed 6 new antibodies to cardiac troponin I. The new antibodies are specific to epitopes 28-34 (MAb Y303), 39-54 (MAb Y309), 161-178 (MAb Y504), 174-191 (Mab Y505), and 182-192 (MAbs Y601 and Y603).
The new clones can be found under Cat.# 4T21cc and RC4T21. All the antibodies can be used for quantitative cTnI immunoassay development. Recommended pairs for the new clones is presented in the table 1.
Table 1. Capture-detection pairs for the newly launch anti-cTnI clones. 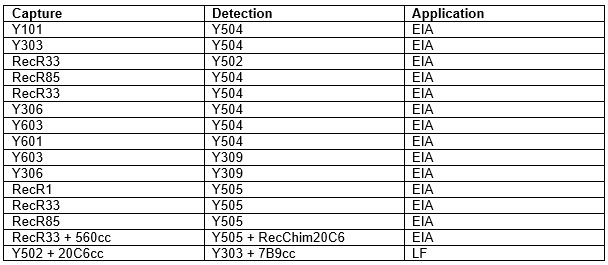
The new clones show very good correlation with existing commercially available cTnI assay (Figure 1). The new clones can be used in several applications, including fluorescence, chemiluminescence (CLIA), and lateral flow platforms (LF). 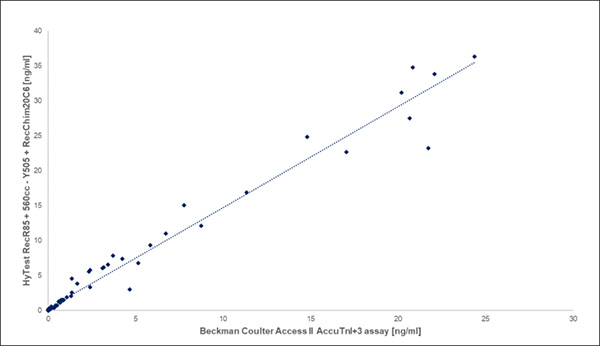
Figure 1. New HyTest anti-cTnI clones shows good correlation (R2 = 0.97) with our existing anti-cTnI clones when compared to an existing commercially available cTnI assay.
Learn more:
Cat# RC4T21: Recombinant anti-cardiac troponin I (cTnI)
