Procalcitonin (PCT) remains one of the most widely used biomarkers for bacterial infection and sepsis management, particularly in guiding antibiotic treatment decisions, including discontinuation. Although PCT assays are widely adopted, global assay standardization remains limited, and assay performance depends heavily on antibody selection and calibration strategy. To support diverse assay formats and development goals, Hytest offers a comprehensive and flexible portfolio of PCT products.
PCT is a 116-amino-acid peptide derived from the 141-amino-acid pre-procalcitonin precursor and can be processed into N-terminal procalcitonin, calcitonin, and katacalcin.
In the diagnostics market, the BRAHMS PCT KRYPTOR method has become an important benchmark for PCT assay development. Reflecting this general assay design logic, PCT immunoassays commonly employ antibody pairs directed against different regions of the PCT molecule, such as N-terminal + calcitonin or calcitonin + katacalcin.
Hytest’s PCT monoclonal antibodies (MAbs), available under Cat. # 4PC47 and Cat. # 4C10cc, have demonstrated strong analytical performance in internal evaluations and provide customers with flexible pairing options targeting different regions of the PCT molecule (see Table 1). They have been validated on Hytest’s in-house chemiluminescence immunoassay (CLIA) and lateral flow (LF) platforms, and have also received positive feedback from customers developing immunoturbidimetric (TIA) assays.
Key advantages of Hytest’s PCT MAbs include:
In addition, Hytest’s polyclonal antibody PPC3 remains an important component of the PCT portfolio and has demonstrated reliable performance in immunoturbidimetric assays. (Availability is limited to selected regions.)
Table 1. Pair recommendations based on different pairing formats and assay platforms
| Intended platforms | Pair recommendations (Capture – Detection) | Antibody pairing formats |
| CLIA | P139/P141 – P135/P223 | Calcitonin + N-terminal |
| P413 – P141 | Katacalcin + Calcitonin | |
| P413 – P223 | Katacalcin + N-terminal | |
| LF | 16B5cc/RC16B5 – P123 | Calcitonin + N-terminal |
| 16B5cc/RC16B5 – PPC3 | Calcitonin + Full length procalcitonin | |
| TIA | 16B5cc/RC16B5 + P123 | Calcitonin + N-terminal |
| PPC3 only | Full length procalcitonin |
Recombinant PCT Antigen Recognized as Reference Material
Hytest also offers recombinant PCT antigen Cat. # 8PC5, suitable for use as a calibrator or reference material. Importantly, 8PC5 has been selected by the Chinese National Institute of Metrology (NIM) as a reference material for procalcitonin. This recognition highlights its strong immunoreactivity, stability, and suitability as a representative material for assay calibration and standardization work.
By combining well-performed monoclonal antibodies, a proven polyclonal option, and a NIM-recognized recombinant antigen, Hytest helps customers develop reliable PCT assays tailored to their platforms and intended clinical use. For more detailed information, you could review PCT related marketing materials, or contact our Sales teams.
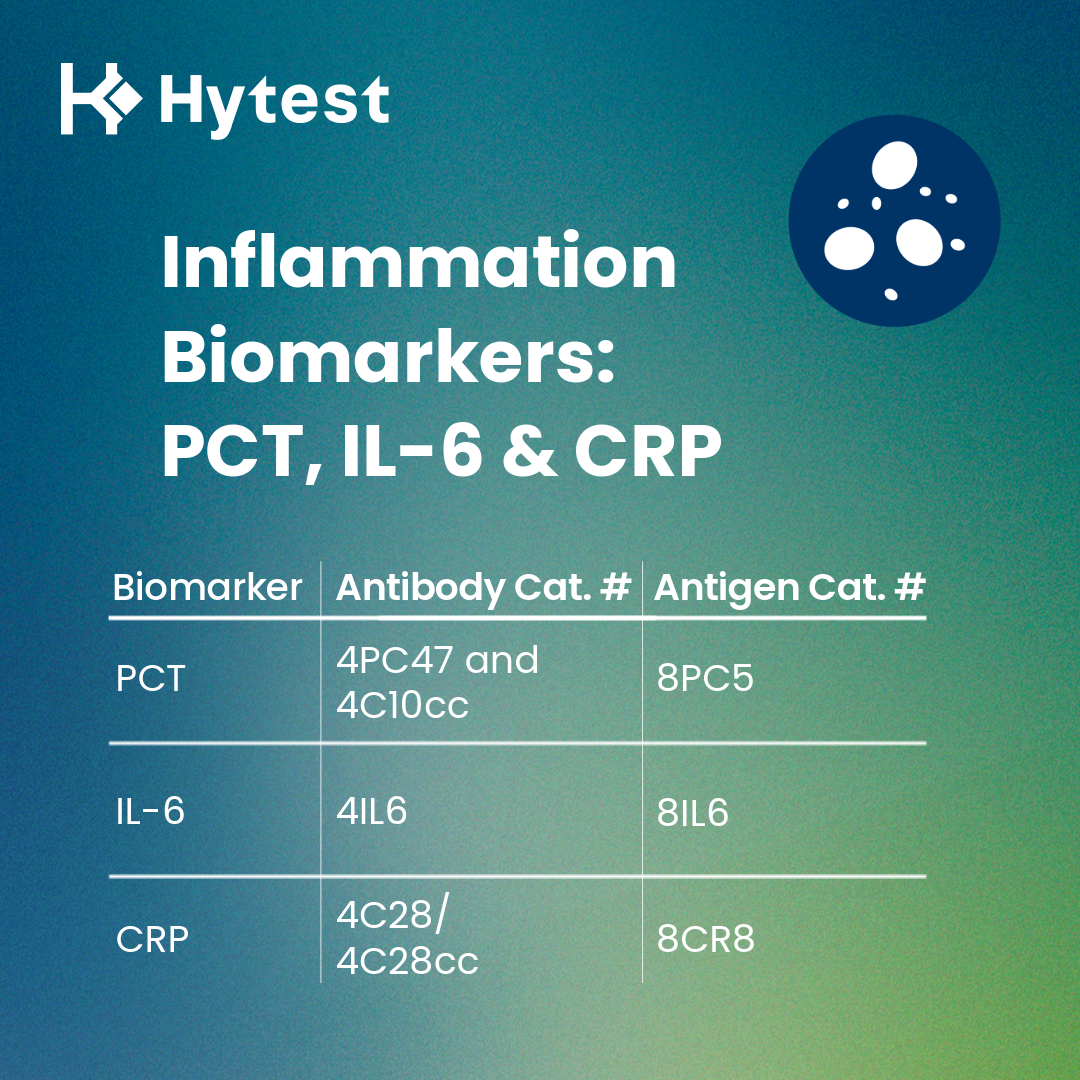
Finally, while PCT is a powerful marker for supporting antibiotic decisions in suspected bacterial infection, clinical interpretation is often strengthened by placing PCT within the broader context of systemic inflammation through the use of IL-6 (Cat. # 4IL6, Cat. # 8IL6) and CRP (Cat. # 4C28/4C28cc, Cat. # 8CR8). Hytest provides antigens and antibodies for both biomarkers, enabling the development of comprehensive multi-marker assay panels.
Read more:
PCT TechNotes